The major difference from electrons is their positive charge. Positrons are the antiparticles of electrons. Therefore, a positron can simply be considered an electron having positive unit electrical charge. It has all the properties of an electron except for the polarity of the electrical charge, which is positive. A positron is the antiparticle of an electron. What is the difference between a positron and an electron?ġ.6. What is a positron in simple terms?ĭefinition of positron : a positively charged particle having the same mass and magnitude of charge as the electron and constituting the antiparticle of the electron. It is a form of antimatter because, when a positron encounters an electron, the two completely annihilate to yield energy. What is a positron simple definition?Ī positron is a particle of matter with the same mass as an electron but an opposite charge. What is another name for electron capture?Įlectron capture is sometimes called inverse beta decay, though this term usually refers to the interaction of an electron antineutrino with a proton. 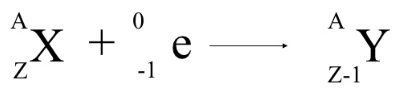
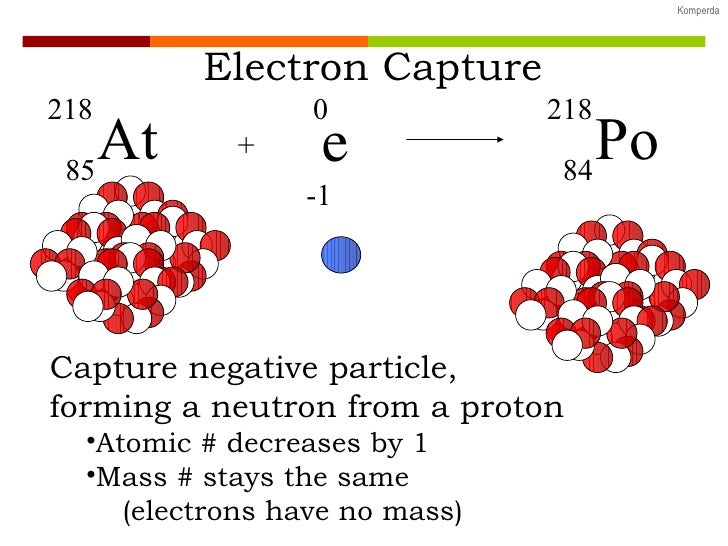
This process will reduce the atomic number by one and not changed the atom’s mass. The result is that a proton will combine with this electron and a neutron is formed. What is the charge of electron capture?Įlectron capture occurs when an inner-orbital electron (negatively charged) is captured by the nucleus (positively charged). In the late 1950s Lovelock was a young researcher attempting to determine the damage done to living cells when frozen. The electron capture detector (ECD) was created by James Lovelock, who built the prototype at his kitchen table. In electron capture, an electron orbiting around the nucleus combines with a nuclear proton to produce a neutron, which remains in the nucleus, and a neutrino, which is emitted. What particle is emitted in electron capture? The mass number does not change, but the atomic number decreases by one during electron capture. The electron on the left side of the equation is usually absorbed from the K or L shell of the parent nucleus.Įlectron capture is the form of radioactive decay that occurs when a nucleus assimilates an electron from an inner orbital. The nuclear reaction depicting electron capture decay is: z. 
What is electron capture formula?ĭetails. This process reduces the atomic number by 1 and emits gamma radiation or an x-ray and a neutrino. Why are the electron capture?Įlectron capture is a type of radioactive decay where the nucleus of an atom absorbs a K or L shell electron and converts a proton into a neutron. In positron emission, a proton inside the radioactive nucleus is converted into a neutron while releasing a positron in electron capture, a proton-rich nucleus of a neutral atom absorbs an inner shell electron which then converts a proton into a neutron, emitting an electron neutrino. The daughter nucleus may be in an excited state. The electron reacts with one of the nuclear protons, forming a neutron and producing a neutrino. 24 What are negative particles called? What happens in electron capture?Įlectron capture is a mode of beta decay in which an electron – commonly from an inner (low-energy) orbital – is ‘captured’ by the atomic nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
